The Platform
One platform for the full ePI lifecycle.
NASHRA gives pharmaceutical teams a single, AI-assisted workspace to manage every stage — from document ingestion to FHIR bundle submission. No more tool-switching. No more chaos.
app.nashra-platform.com/dossiers
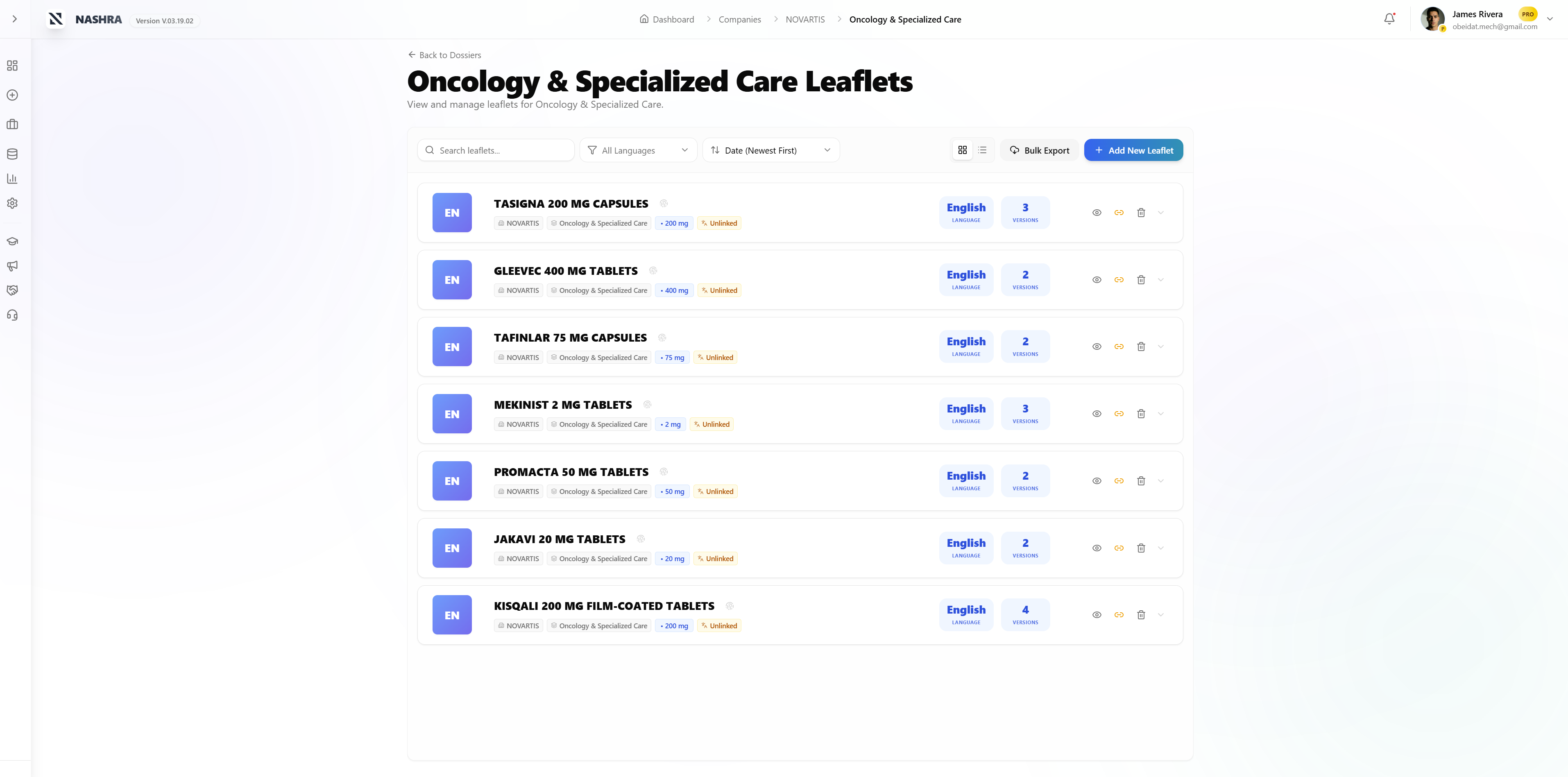
Portfolio Hierarchy
Total control. Zero chaos.
NASHRA structures your entire pharmaceutical portfolio in a logical, auditable hierarchy — from the marketing authorization holder down to individual version diffs.
MAH
Marketing Authorization Holder
Dossier
Product / Market Authorization
Leaflet
PIL / SmPC / Package Insert
Version
Full audit trail + diff tracking
Module Breakdown
Four modules. One complete view.
General Module
Product name, authorization number, MAH details, regulatory status.
Organization Module
Manufacturer, distributor, and authorization holder records.
Medicinal Module
Active substances, excipients, clinical particulars, pharmacology.
Composition Module
Qualitative and quantitative composition. FHIR Ingredient resources.
Everything Included
Built for the full workflow.
- PDF / DOCX → AI-extracted structured content
- MAH → Dossier → Leaflet → Version hierarchy
- FHIR R5 bundle generation with one click
- Built-in NeuroTVT cross-reference auditing
- Full version inheritance and diff tracking
- Arabic / English bilingual FHIR support
- Row-level security — enterprise grade
- Real-time collaboration for regulatory teams
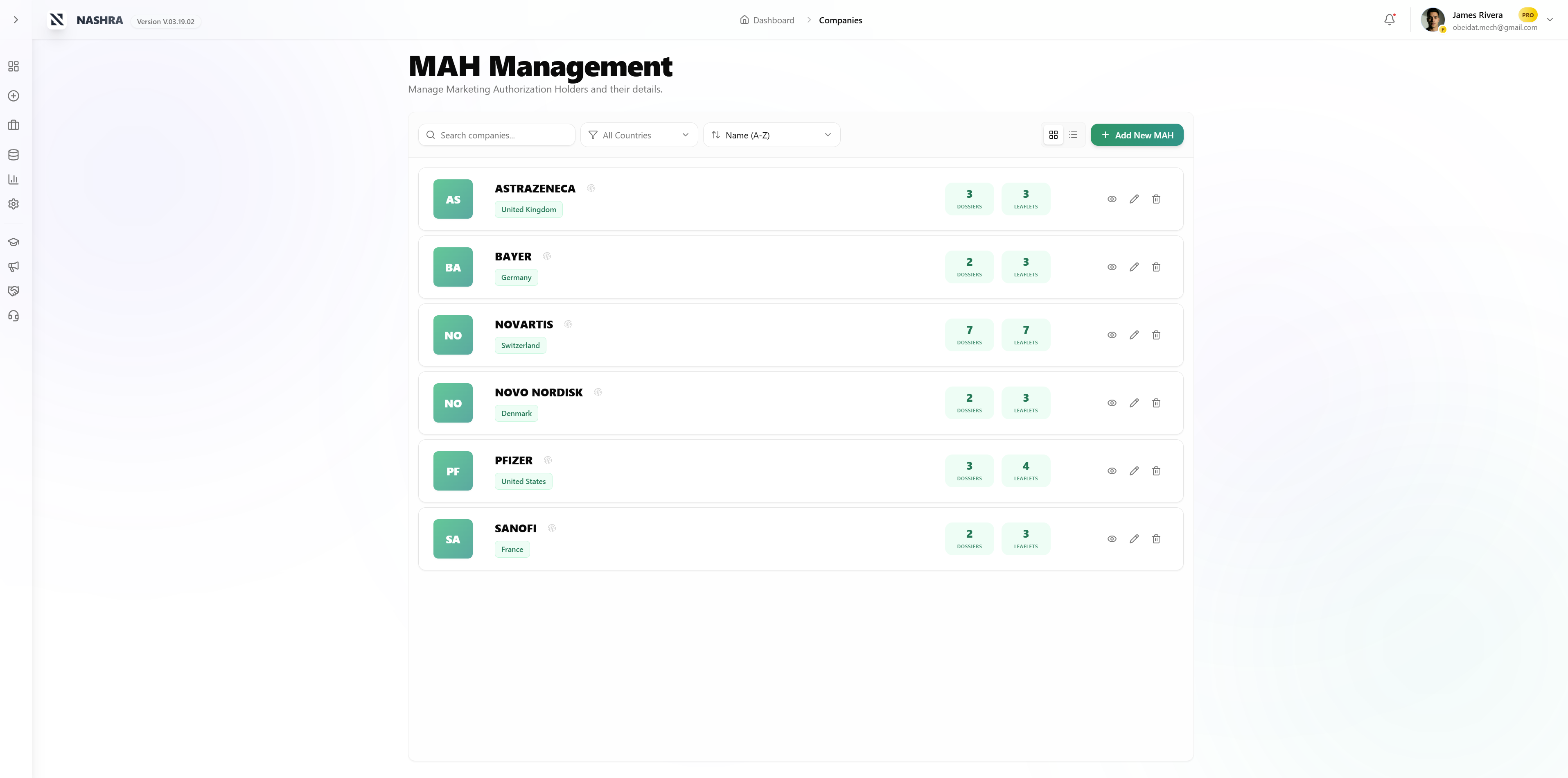
See it in action.
30 minutes. No commitment. We'll walk through your exact document type.
Request Access